 This is a guest post by Arif Ashraf.
This is a guest post by Arif Ashraf.
Arif Ashraf is PhD student at Iwate University, Japan and Graduate Student Ambassador of ASPB. His research interest is understanding the hormonal interplay in primary root development of Arabidopsis. He blogs about plant science at http://www.aribidopsis.blogspot.com
Like us, plants have stem cells too. These are innate, undifferentiated cells localized in both root and shoot meristems. They house precursor cells that divide, elongate/expand, and finally form differentiated tissues and organs of plants. It’s how plants generate the roots, trunks, branches, leaves, flowers, and all the other structures we witness as the green world.
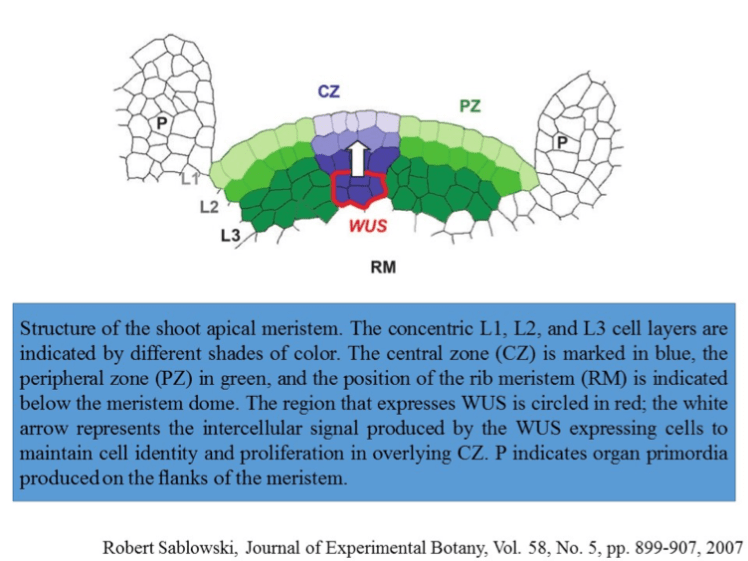
The shoot apical meristem (SAM) is a complex structure consisting of three distinct layers (denoted L1, L2 and L3) in the dicot (one of the two major groups of flowering plants) Arabidopsis thaliana. L1, L2 and L3 form the epidermis, sub-epidermal tissues, and inner tissues of the shoot, respectively [1]. Cells in the L1 and L2 layer divide in a sideways fashion, keeping these layers distinct, whereas the L3 layer divides in a more random fashion.
Apart from the cell layers, the SAM is divided into three dynamic functional zones/cell populations. The central zone (CZ) is maintained through a low rate of cell division and supplies cells for the peripheral zone (PZ) and the rib system (see the figure below). The (PZ) is responsible for the formation of lateral organ primordial like leaves (P), whereas the rib meristem (RM) pushes the SAM upward. A group of cells residing directly below the stem cells in the CZ is referred to as the organizing center (OC) [2].
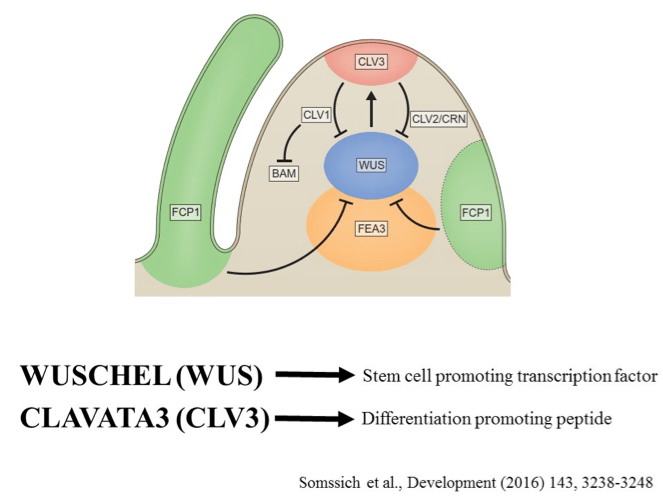
Shoot meristems are maintained by pluripotent (unspecialized with potential to become any cell type) stem cells controlled by CLAVATA-WUSCHEL feedback signaling. This pathway, which coordinates stem cell proliferation with differentiation, was first identified in Arabidopsis, but appears to be conserved in diverse higher plant species including crops. The CLAVATA3 (CLV3) – WUSCHEL (WUS) signaling pathway has evolved as the central regulatory pathway that coordinates stem cell proliferation and differentiation. This coordination is achieved via an auto-regulatory negative-feedback loop comprising the stem cell-promoting transcription factor WUS and the differentiation-promoting peptide CLV3. CLV3 – WUS maintain a circuit, where CLV3 is only expressed in stem cells of SAM and perceived by receptor like proteins in OC and repress WUS expression. Due to the repression of WUS, few stem cells are maintained and reduction in CLV3 production [3].
To understand the function of a gene, scientists usually mutate that particular gene leading to loss-of-function (LOF – i.e. “breaking a gene”) or gain-of-function (GOF – i.e. “making more of a gene or making it more effective than usual”) phenotype of plants. Based on our knowledge about CLV3 – WUS circuit combined with mutant studies, take a look at SAM development of a crop plant, maize (Zea mays).
Through mutagenesis screening, fasciated ear 3 (fea3) mutant has been identified which contains a larger SAM like clv mutant. FEA3 is a CLV-type LRR (Leucine Rich Repeat) receptor-like gene localized to the plasma membrane. So, it was predicted that exogenous application of CLV3 would reduce SAM size. They applied CLV3 peptide to two mutants (fea2, fea3) and found that fea2 is resistant for meristem growth, but not fea3. It tells us that FEA2 perceives CLV3, not FEA3 [4].
The next idea was that FEA3 perceives something other than CLV3. According to phylogenetic analysis, three similar genes (ZmCLE7, ZmCLE14 and ZmFCP1) to CLV3 were identified in Zea mays. When these three peptides are applied exogenously on wild type plants, they reduced root growth. But, when these three peptides are applied exogenously on fea3, ZmFCP1showed resistant for root growth and SAM size also. Additionally, Zmfcp1 mutants also show fasciated ear phenotype and suggests that FEA3 – FCP1 pathway is regulating SAM signaling from differentiating cells [4].
Although CLV – WUS system is conserved from model plant Arabidopsis to crop plants, buffering of this circuit is more prominent in Arabidopsis than in crop plants. Even weak mutants of this circuit show significant phenotypes, because crop plants went through selection based on yield during domestication. Over the years, farmers selected the varieties containing increased kernel row number, kernel numbers per ear, and ear weights without considering the underlying mechanism. Comprehensive molecular biology and phenotypic study of fea3 mutant showed that high yielding variety is actually a reflection of a manipulated CLV – WUS circuit [4].

This study from Je et al. [4] has shown the promising impact of basic research to translate into an applied field such as developing high yielding crops. At the same time it suggests a universal pathway to control cell differentiation during plant development.
References:
- Satina, S., A. Blakeslee, and A.G. Avery, Demonstration of the three germ layers in the shoot apex of Datura by means of induced polyploidy in periclinal chimeras. American Journal of Botany, 1940: p. 895-905.
- Sablowski, R., Flowering and determinacy in Arabidopsis. Journal of Experimental Botany, 2007. 58(5): p. 899-907.
- Somssich, M., et al., CLAVATA-WUSCHEL signaling in the shoot meristem. Development, 2016. 143(18): p. 3238-3248.
- Je, B.I., et al., Signaling from maize organ primordia via FASCIATED EAR3 regulates stem cell proliferation and yield traits. Nature genetics, 2016.